Company News
News
Important Update: GasD IS Intrinsic Safety Certification Update
We are pleased to share an important progress update regarding the intrinsic safety certification of the GasD IS portable gas detector. All intrinsic safety testing has been successfully completed. UL’s evaluation confirms that the GasD IS design meets the requirements of IEC 60079-11 for intrinsic safety. The certification process has now moved into its final administrative and documentation phase ahead of formal approval. Read more here about Important Update: GasD IS Intrinsic Safety Certification Update...
News
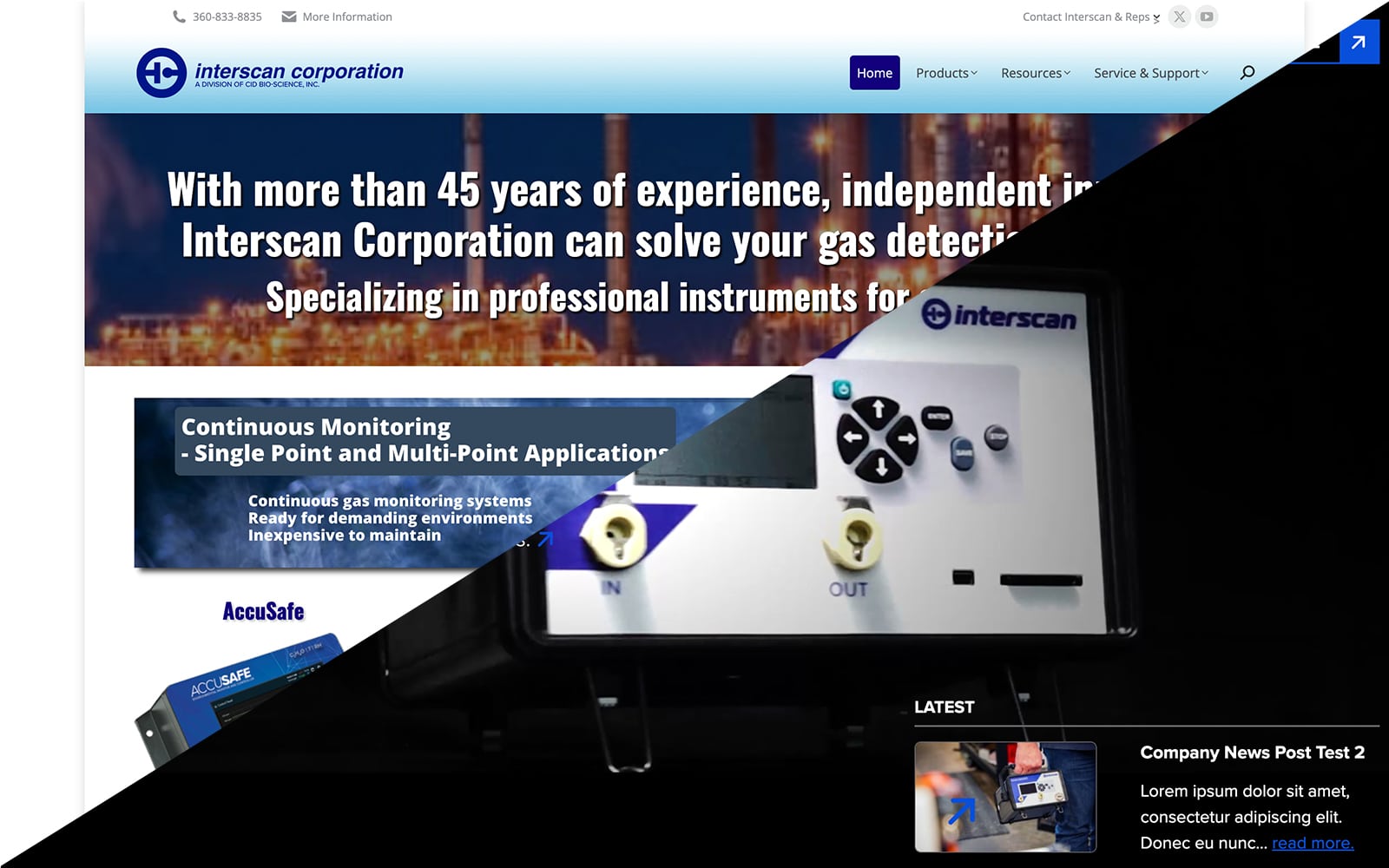
Interscan Corporation Unveils Redesigned Website
Interscan Corporation is thrilled to announce the launch of our newly redesigned website, going live on Friday, May 17, 2024. The new design reflects our commitment to improving user experience, focusing on making our site more modern, user-friendly, and informative.
Old site:
New Site:
Key Improvements:
Streamlined Navigation: Our new website features a cleaner, more intuitive layout, making it easier for visitors to find the information they need. Read more here about Interscan Corporation Unveils Redesigned Website...
News
Celebrating Milestones the CID Bioscience Way: A Waffle Day to Remember
In the heart of Camas, WA, something special took shape in the halls of CID Bioscience. The scent of success? No, it was the sweet, mouthwatering aroma of freshly made waffles! Not your average corporate celebration, but at CID Bioscience, we believe in doing things a little differently.
What’s the cause of the celebration? Our unique culture thrives on innovation, teamwork, and an appreciation for good waffles. Read more here about Celebrating Milestones the CID Bioscience Way: A Waffle Day to Remember...